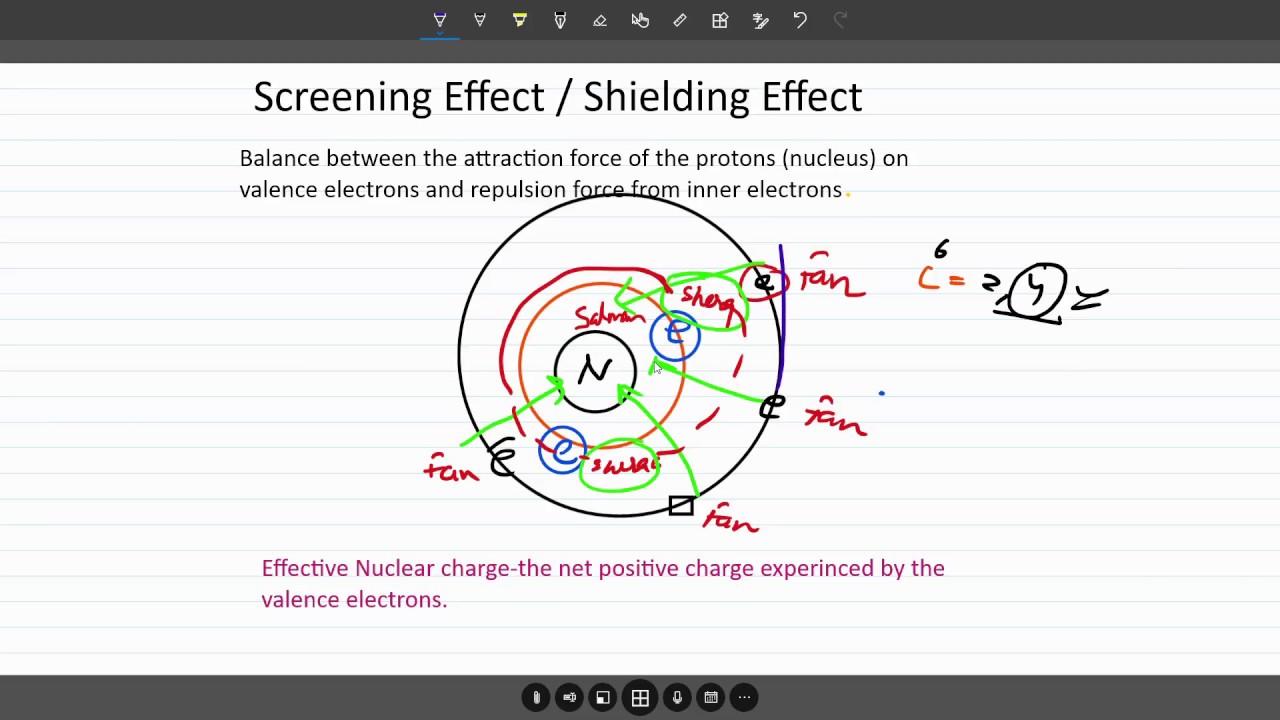
According to I.C slater effective nuclear charge, Z^(**), due to screening, is not exactly equal to the actual nuclear charge Z of the nucleus of the atom. Z^(**) depends on the type
54. In the lithium atom screening effect of valence shell electron is caused by 1) Electron of k and L shell 2) Electron of k shell 3) Two electron of 1st and
![PDF] Electron screening and excitonic condensation in double-layer graphene systems | Semantic Scholar PDF] Electron screening and excitonic condensation in double-layer graphene systems | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/12258383aed45faa8f3fb8f9277a44ffb3624dd7/1-Figure1-1.png)
PDF] Electron screening and excitonic condensation in double-layer graphene systems | Semantic Scholar

SOLVED: Question 37 (1 point) Screening of the nuclear charge by core electrons in atoms is: less efficient than by valence electrons more efficient than by valence electrons essentially identical to that

PDF) Electron screening effect in the reactions 3He(d, p) 4He and d( 3He, p) 4He Supported in part by INFN, BMBF (06BO812), DFG (436UNG113-146) and OTKA (T025465 | Matthias Junker -

According to I.C slater effective nuclear charge, Z^(**), due to screening, is not exactly equal to the actual nuclear charge Z of the nucleus of the atom. Z^(**) depends on the type